Nitrogen is an essential component of everything that lives and moves on Earth. Without nitrogen, no amino acids or nucleic acids, no metabolism or proteins. The human body contains approximately 7% nitrogen, making it the fourth most abundant element in the body. After oxygen, carbon and hydrogen. About 78% of the Earth’s atmosphere is made up of nitrogen, 21% of oxygen, and the rest of various other noble gases. Nitrogen is therefore present in large quantities in the air. We do not use the nitrogen that we inhale as humans and we also exhale it. However, this nitrogen is very useful for various industrial applications, and more specifically in the transport sector.
Nitrogen, chemically indicated with the symbol N (nitrogen gas N2), gets its name from the fact that it can suffocate life and fire. In high concentrations, nitrogen has an oxygen-repelling effect. We recover nitrogen by fractionally distilling the air. i.e. We split the air and extract the nitrogen. The freezing point of nitrogen is -210°C and the melting point -196°C at normal atmospheric pressure. This means that at temperatures below -210°C, nitrogen is a solid. This occurs in practice, but less often than the liquid or gaseous form. It is liquid between -210°C and -196°C. Nitrogen gas is obtained above -196°C. A widely used gas in industry. It is colorless and odorless and, unlike oxygen, an inert gas.
Research shows that more than sixty percent of motorists and by extension the transport sector drive with tires that have the wrong pressure. Usually the pressure is not compliant. A tire with ‘regular air’ quickly becomes five to ten percent softer over time. Oxygen molecules are so small that they escape through the tire carcass. Nitrogen molecules are larger and hardly escape, if at all. Tires that are and remain inflated wear less quickly and ensure that rolling resistance remains optimal. That results in lower fuel consumption. An underinflation of twenty percent causes a thirty percent faster loss of efficiency of the tire. Thirty percent undervoltage even results in an average 5% higher fuel consumption. You can recognize nitrogen in tires by the green valve cap that is used.
Nitrogen in the tires has a large number of advantages, which are achieved because the tires stay inflated longer. Tires with nitrogen lose pressure on average 4 times less quickly than tires with air. It is recommended, for tires filled with air, to check the pressure once a month. For tires with nitrogen, 4 times a year is sufficient. In addition to this advantage, there are a number of other advantages with regard to costs and safety. Below you can read about these advantages: The molecules of nitrogen are larger than those of air. Because rubber is never 100% airtight, molecules escape through the tire and the tire pressure decreases. Because oxygen molecules are smaller than nitrogen molecules, you can imagine that this process goes faster and the tire pressure drops faster. Because inflation with nitrogen achieves a maximum amount of 99% nitrogen, the difference seems minimal. As with all substances, nitrogen is subject to the rules of expansion, which occurs during temperature rise, and condensation, when cooled. In practice, for every temperature increase of 8°C there is a pressure increase of 0.1 bar and vice versa. The use of nitrogen keeps the temperature in the belt more constant. In this way, the tires wear more evenly and the rolling resistance is optimal. Rolling resistance corresponds to the energy lost when the tire bulges. The energy loss is due to the continuous material deformation of the belt. The right voltage leads to less energy loss. Oxygen reacts in tires with rubber, water particles and dirt, causing rust on the rim and deterioration of the rubber. Nitrogen stops these oxidation processes The inside of tubeless tires is provided with a tubeless layer that should make the tire as airtight as possible. However, air molecules slowly but steadily continue to escape through this tubeless layer: this natural process is called permeation. Nitrogen molecules are larger than oxygen molecules, making it more difficult for nitrogen molecules to pass through this tubeless layer: eventually both will find their way through the tubeless layer, but it takes nitrogen molecules up to 6 times longer than oxygen molecules.
With tires at the right pressure, your car uses less fuel. Because the tires stay inflated longer with nitrogen, fuel consumption is lower. On average, filling with nitrogen results in savings of 5%. Less rolling resistance and therefore less unnecessary fuel consumption.
Another advantage of keeping your tires inflated for longer is that wear is much lower. A tire pressure of 20% lower than recommended results in increased tire wear of 30%. Or in the opposite direction a direct saving of 30% on tires.
Nitrogen has been used in the aircraft industry and Formula 1 for many years. An important reason to use nitrogen for aircraft tires and Formula 1 tires is the fact that nitrogen has the (pleasant) property of counteracting heat development. Because nitrogen in the tire is a homogeneous mixture, expansion is much less; i.e. the temperature of ordinary air with oxygen increases sharply, especially with prolonged and heavy use. In the transport sector, long-term and heavy use of tires is self-evident. The use of nitrogen prevents heat development, so that the lower temperature ensures fewer tire problems, such as tread separation and punctures. As a result, there is less chance of a breakdown on the road, which saves a lot of money. One would think that all trucks and trailers are equipped with nitrogen, but in reality there is still a lot of work to be done here. Otherwise, in the aviation sector, money is an obligation to use nitrogen. One reason why airplanes use it is that, because there is no moisture in nitrogen, the tires cannot freeze during the flight, but are also more resistant to sudden contact with the concrete runway during landing. In Formula 1 and racing cars, nitrogen is always supplied. Think, for example, of the 24 hours of Le Mans, where the endurance of all parts of the car comes down to endurance, so it makes sense that nitrogen is used here. Also the advantage for tires on collector cars, which are used less often, so that the tires tend to deform permanently. But also for campers and caravans Recently, there has also been an awareness among private car owners. The cost of a nitrogen-filled tire does not outweigh the disadvantages of ordinary compressed air.
Ordinary air is freely available. Nitrogen must be specially purchased in gas cylinders, or made using a nitrogen generator. Both involve an investment that must be passed on. Filling tires with nitrogen is therefore by definition more expensive than compressed air.
But this cost does not outweigh the many benefits.
When your tire pressure is lower, your braking distance becomes longer and the chance of skidding is greater. Nitrogen heats up less quickly than air. This has the advantage that the tires heat up less and therefore the chance of a blowout is smaller. Most blowouts are caused by a combination of heat, age, water, corrosion, and incorrect tire pressure.
Air contains hydrogen, which causes condensation to form in the tire and rim. This causes the metal to oxidize (this applies to both alloy rims and steel rims). Oxidation of the metal allows air to escape between the tire and rim sooner, causing the tire pressure to drop faster and faster in the long term. . Other advantages of using nitrogen in tires are better and more consistent driving comfort and better handling. The use of nitrogen therefore also improves road safety. For example, the chance of overheating a tire that is at the right pressure is much smaller. Tire pressure can also increase by using standard compressed air. A final advantage of nitrogen over air is that no more oxygen and water vapor enter the tire. This makes condensation in tires a thing of the past, which prevents rusting on rims. Rust is again one of the causes of air escaping from tires. Nitrogen has a homogeneous composition, which means that it expands less at high temperatures. This reduces the risk of a tire blowout at high or overloaded conditions. Nitrogen is also absolutely not dangerous. Nitrogen is colorless, odorless, non-toxic and cannot burn. There is no additional danger in the event of a blowout. Blowouts do not occur very often, especially when the tire is filled with nitrogen. When a tire with nitrogen filling bursts, nothing else happens than when a tire with regular air (80% nitrogen, 20% oxygen) bursts. There is certainly an advantage of nitrogen in the event of a fire hazard. If, for example, a jammed brake causes a fire to start and the tire bursts, the fire will be extinguished rather than fanned. A fire needs oxygen. Due to the dose of nitrogen in the tire, this will neutralize the fire, in contrast to oxygen, which actually fuels a fire. Finally, unlike oxygen, nitrogen is not flammable. This is especially beneficial when you transport dangerous goods. An additional advantage is that the steel cord layers of the tire will never be affected, which is beneficial for tires that are retreaded, and here too the risk of a blowout is greatly reduced.
A nitrogen-filled tire is ‘clean and dry’. Nitrogen, unlike oxygen, is non-flammable. In case of fire accidents, nitrogen-filled tires even have an extinguishing effect.
Compressors, even in tire centers and in many transport companies, are not always equipped with a condenser dryer (note that so-called water separators have a very minimal effect) that should remove most of the moisture from the compressed air. As a result, the compressed air contains a lot of water vapour, especially when the ambient air is warm (summer). This water vapor has 2 adverse effects:
Moisture in the compressed air accelerates the occlusion of valves, TPMS sensors and rims
Moisture in the compressed air amplifies pressure variations due to temperature fluctuations. Every gas expands when it gets hot, and contracts when it cools: ordinary air has a deviation of 1 PSI per 12 degrees Celsius. Tires heat up from the ambient temperature, the sun, and driving: a decent ride can heat up your tires by up to 80°. It is therefore always recommended to check the tire pressure ‘cold’ (in the morning, before departure).
Inflating with nitrogen with a purity of 99% ensures a reduction of the natural pressure compared to air containing oxygen (without external influence). In addition, nitrogen is a stable type of gas that reacts less to temperature increases caused by rolling resistance. Among other things, the tires can handle high speeds on circuits without affecting power; in short, in every situation this improves the performance and lifespan of our expensive tires.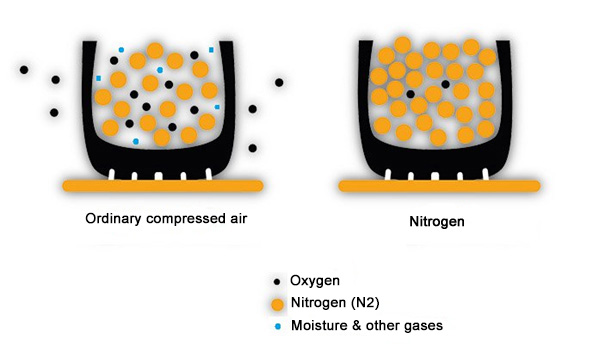
Suppose you have nitrogen in the tires but notice that your tire has too little pressure, can you top it up with regular air? Yes, that’s possible. Of course mixing reduces the effect of nitrogen, but it is certainly possible. It is recommended to always fill the tires with nitrogen and to go to a tire specialist if the pressure is too low. A tire center that respects itself a little will almost always be able to fill your tires with nitrogen.
Filling your tires with nitrogen has a positive impact on the environment. This is due to the aforementioned benefits of lower fuel consumption. You can imagine that when more and more people have tires filled with nitrogen and therefore have lower fuel consumption, this is positive for the environment. For cars and trucks, both summer tires and winter tires are supplied with nitrogen.
As a rule, there are 2 options for using nitrogen for filling tires. On the one hand, relatively expensive and hardly controllable filling is still used, provided that steel high-pressure cylinders are filled with nitrogen. However, if one wants to approach this professionally and certainly to transport companies and tire centres. Is a nitrogen generator fed by a compressor with dryer the ultimate solution. Even though one truck tire contains about 20 times as much nitrogen than a passenger car tire and the desired pressure is much higher, a nitrogen generator with the necessary programming will be the solution. Javac offers 3 different nitrogen generators depending on the application, so a generator designed for car tires will not suffice for truck and bus tires, for example.
Otherwise you can. In principle, Javac supplies tire filling systems in every requested capacity.
A filling system is extremely interesting for a tire center as well as for small and large transport and bus companies and fleet owners. It suffices to point out to the customer who comes to replace his tires that the life of his tires will increase considerably, and certainly already now because our fleet has the tendency to electrify, which means that the wear of tires due to the heavy batteries and high acceleration speeds are significantly higher than conventional cars. It is no longer even responsible for transport companies to fill the tires with normal compressed air, count on the tires lasting up to 35% longer, blowouts, and expensive interventions, for example. be avoided. A good filling installation can pay for itself within 6 months. The technical lifespan is of course many times longer. The nitrogen generator is available with 1 or more filling hoses. On average, it takes 3 minutes to fill a truck tire. You should preferably have a (screw) compressor with an operating pressure of 12 bar and a flow rate of 1500 litres/minute, and a condenser dryer. In most cases, the company is already equipped with this compressed air installation. The nitrogen generator is connected to a galvanized vessel of at least 500 litres. Contrary to what is supposed, replacing the filters is very affordable. Depending on the intensity of use and the quality of the ambient air, and the filters present on the compressor used, this is between 6 months and 24 months. The PLC on the generator indicates with a signal when the filters need to be replaced. The same 2 filter cartridges are used for all our versions.
The cost is 75 Euro per set. This cost is almost negligible per tire filling.
Never use a vacuum pump to prepare your tires for using nitrogen, as this can seriously damage the internal structure of your tires.
If you have any questions, don’t hesitate to contact.
What can we do for you?
If you are looking for the alternative with an unprecedented price/quality ratio.
Then we may be your new point of contact, we are ready, are you?